By Torsten Liem & Miriam Havel
Summary
In this article, the importance of the suprachiasmatic nucleus (SCN) for the diurnal rhythmic regulation of the intrinsic time programme of living organisms is presented and clinical references to disorders of the SCN are shown. Via the numerous neuronal and neuroendocrine connections, the SCN integrates temporal information from the environment on the one hand and the endogenous biological rhythms of neurohormonal axes - (hypothalamic-pituitary-adrenal axis (HPA axis), hypothalamic-pituitary-gonadal axis (HPG axis), thyrotropic regulatory circuit, etc.) - on the other. Via these and other complex interaction pathways, SCN regulates the neurovegetative system as well as metabolic, immune and behavioural functions. Clinically relevant results for osteopathic treatments are discussed.
Keywords
Environmental stimulus, circadian rhythm, hypothalamic-pituitary-adrenal axis, hypothalamic-pituitary-gonadal axis, neurohormonal axis, pituitary-thyroid regulatory circuit, metabolic regulation, autonomic nervous system, neurvegetative
Abstract
This article elucidates the role of the suprachiasmatic nucleus (SCN) in the circadian regulation of the intrinsic temporal programme of living organisms and depicts some clinical aspects of its misalignment. Due to its numerous neuronal and neuroendocrine connections, the SCN integrates temporal information of the environment on the one hand and the endogenous biological rhythms of neurohormonal axes - hypothalamic-pituitary-adrenal axis (HPA-axis), hypothalamic-pituitary-gonadal axis (HPG-axis), thyrotropic feedback control etc. - on the other hand.
Through these and other complex interaction pathways SCN regulates the neurovegetative system as well as functions of metabolism, immune system and behaviour. Clinical aspects relevant for osteopathic treatments are discussed.
Keywords
Environmental stimulus, circadian rhythm, hypothalamic-pituitary-adrenal axis, hypothalamic-pituitary gonadal axis, neurohormonal axis, metabolic regulation,
autonomic nervous system, neurovegetative system
The suprachiasmatic nucleus (SCN)
Life on earth has developed in the context of rhythmic environmental stimuli. The temporal organisation in behaviour (food intake, sleep, activity) and in physiology is common to all organisms and reflects the rhythm of the cyclical rotational movement of the sun, moon and earth. Corresponding to spatial orientation, which requires an internal representation of the outside world, an internal representation of these time periods is necessary for meaningful orientation in the temporal space of a day, month and year. Temporal coordination is subject to control by an endogenous time programme and requires a predictable and repetitive rhythm in phylogenesis (Dumbell et al. 2016).
In mammals, this endogenous time programme finds its morphological correlate in the suprachiasmatic nucleus (SCN) of the hypothalamus. This paired neuron group of (in humans) around 20,000 densely packed nerve cells in the anterior hypothalamus acts as a so-called central circadian pacemaker or "master-clock" the entire internal rhythm of the organism. In mammals, the internal rhythm is organised hierarchically by the SCN, which regulates and synchronises the peripheral oscillators in all other tissues as well as our sense of time through endocrine and neurovegetative signals (Dibner et al. 2010).
Function of the suprachiasmatic nerve
General function of the suprachiasmatic nucleus
Circadian rhythm by means of light-dark alternation via retina-hypothalamic projections (→ corpus pineale)
- Wake/sleep.
- Circadian rhythm-controlled activity of the endocrine, neurovegetative, behavioural (food intake, sleep, activity), metabolic and immune systems, body temperature, blood pressure, etc.
The search for the clock
The possibility of making targeted lesions on various brain structures in animal experiments led to the more precise localisation of the endogenous pacemaker in mammals. Curtis Richter, who first established the well-known hamster wheel for the standardised investigation of spontaneous activity phases of small rodents in the laboratory, discovered after numerous meticulous experiments in the 1950s and 1960s that rats lost their rhythmic behaviour after lesions in the rostral area of the hypothalamus (Richter 1967). Moore and Eichler suspected a close anatomical-functional relationship to the visual system and in 1972 identified a projection of nerve fibres leading from the optic nerve to the hypothalamus using nuclear medicine tracer methods (Moore et al. 1972).
At the same time, Stephan and Zucker achieved not only the loss of rhythmic behaviour of food intake and the sleep and wake cycle through experimental lesions of the SCN region of the test animals. The ovulation cycle and rhythmic secretion of corticosterone (the most important corticosteroid in rodents) were also disturbed in the otherwise completely healthy animals (Stephan and Zucker 1972).
In electrophysiological studies with isolated SCN tissue, a synchronous rhythmic electrical activity of the cells with a phase length of approximately 24 hours was demonstrated (Inouye et al. 1979). Explantation and transplantation experiments confirmed that the essential mechanism of biological clocks is expressed at the cellular level. The rhythmic spontaneous activity of SCN neurons in vitro and their metabolic activity, as observed in vasopressin secretion and uptake of radiolabelled 2-deoxyglucose, provided further evidence for the central role of the SCN (Schwartz et al. 1980).
The more or less accidental discovery of two hamsters in an experimental animal population, which showed a free-running spontaneous sleep-activity rhythm of 22 hours in constant darkness (in contrast to a circa dianen(i.e. a daily rhythm of 24.1 hours, as is usual in hamsters in the scientific laboratory) enabled further clarification of the pacemaker function of the SCN. The biologists Ralph and Menaker succeeded in generating this "faster" endogenous sleep-wake rhythm in the "slower" animals using foetal SCN transplants from animals with a day length of 22 hours. The biological rhythm of the arrhythmic rodents, which had previously ablated SCN, was restored and exhibited the circadian characteristics of the donor (Ralph and Menaker 1988). The central pacemaker function of the SCN, which regulates the endogenous time programme, could thus be clarified.
The path of light to the SCN
The synchronisation of the internal clock with the temporal cues of the environment is not a passive reaction, but a complex adaptation process (so-called "entrainment"). In terms of evolutionary physiology, this temporal-organisational adaptation offers organisms the advantage of anticipating environmental conditions and thus optimising their use.
The adaptation process depends on the properties of the endogenous pacemaker and the external stimulus. If there is a lack of external temporal information, as in the case of Ralph and Menaker's hamsters kept in constant darkness, this leads to the manifestation of the free-running periodicity of the endogenous oscillator (in the case of "fast" hamsters, the "days" were 22 hours long).
The outer one, "entrainment" generating Stimulus is referred to as a zeitgeber. A basic distinction can be made between photic (light) and non-photic zeitgebers (e.g. temperature fluctuations, social contact, food intake). In most living organisms, the zeitgeber is represented by a stable environmental signal of the time of day, the alternation of light and dark. For example, from around 4 a.m. every day, the human organism adjusts to the start of the day. Daylight triggers a whole cascade of physiological changes.
The ability of a zeitgeber to adapt a biological system to the circadian rhythm requires that information about the external time of day, month and year reaches the endogenous oscillator. For most living organisms, (sun)light is the most important zeitgeber. A close anatomical relationship to the visual system is therefore obvious. The histological structure of the eye had already been well studied for more than a century, and new anatomical findings were not to be expected when chronobiologists provided increasing evidence of another photosensitive system in the eye (in addition to cones and rods) in the mid-1990s. Using an animal model (mouse), they were able to show that visually blind animals with degenerated, non-functional cones and rods continued to present adequate circadian responses to light stimuli, i.e. they did not suffer from circadian "blindness". When the entire eye and thus also the light receptors for the circadian system were lost, there was no longer a circadian response to light impulses (Lucas et al. 1999). Light intervention studies in humans also showed that blind subjects with externally intact eyes can be synchronised to the external day with the help of bright light (Czeisler et al. 1995).
The light reception relevant for the circadian system takes place at the retina, but not through rods and cones as traditionally assumed. Rather, light travels via specialised, directly light-sensitive retinal cells (so-called intrinsically photosensitive retinal ganglion cells, ipRGCs), which make up around 1% of the entire retinal ganglion cell population and are located in particular in the inferior-nasal/medial area of the retina (Foster et al. 2002). They lead to the SCN with their descending nerve axons via the retinohypothalamic tract (RHT). In purely morphological terms, the RHT is derived from axon collaterals of the axons of the optic nerves, which grow from the optic chiasm into the ventral part of the SCN around the 4th postnatal day (Stephan and Zucker 1972). Light detection of the directly light-sensitive retinal ganglion cells takes place by means of the photopigment melanopsin, which is phylogenetically much older than the photopigments of the cones and rods (Provencio et al. 2002; Sekaran et al. 2003).
The signal transduction cascade that leads to the triggering of the cellular action potential is melanopsin-triggered after the photon-absorbing chromophore molecule 11-cis-retinaldehyde is converted to the all-trans state. Initially identified in the retinal ganglion cells of rodents (which are mostly nocturnal), melanopsin has also been isolated in (diurnal) macaques, whose visual system is similar to that of humans (Dacey et al. 2005). The maximum absorption of melanopsin is at 480 nm, which corresponds to the blue part of the light spectrum. The melanopsin-producing ganglion cells can thus identify the (particularly blue-rich) light of twilight and thus perceive the transition between day and night due to the altered spectral composition of the light (Foster 2005).
In contrast to the visual system, which provides precise information about the spatial and temporal resolution of a light signal ("image forming retinal pathway"), the projection of the circadian system contains information about the intensity and spectrum of the light received ("non-image forming retinal pathway"). These two systems do not work completely separately from each other, as some retinal light information also reaches the SCN from the corpus geniculatum laterale (a brain structure that transmits information from the image forming retinal pathway to the visual cortex) via the tractus geniculohypothalamicus, which suggests an interaction (Foster 2005).
After switching in the paraventricular nucleus and supracervical ganglion, the circadian information is transmitted from the SCN to other brain structures, including the corpus pineale, a hormone-producing gland also known as the pineal gland or epiphysis. The role of this organ, apart from melatonin production, has not yet been clearly clarified; a control function of seasonal rhythmic processes, such as reproduction and hibernation, is assumed (Pevet 2000).
In reptiles and some bird species, the epiphyseal tissue is directly sensitive to light (hence the term "third eye") and represents the dominant rhythmic oscillator that controls the diurnal periodicity (Gaston et al. 1968; Underwood et al. (1990).
Internal synchronisation and paracrine mechanisms within the SCN
Under in vivo conditions, the SCN integrates exogenous and endogenous signals, in particular the light-dark alternation, and thus enables the organism to optimally prepare itself for the environmental conditions. The special feature of the SCN cells is that they maintain their rhythmic activity even without external influences. Autonomous circadian oscillations of neuronal activity have been demonstrated both in explanted SCN tissue and after isolation of individual SCN cells in vitro (Welsh et al. 1995). However, the comparison between SCN tissue cultures and cultures of individual cells shows that the deviations of circadian periods of the cells in the brain slices are smaller (Herzog et al. 2004).
The individual oscillators with quite different endogenous periods stabilise and synchronise each other. The higher the degree of organisation of the cells, the more stable and precise the circadian oscillation. A coherent pacemaker function therefore appears to be dependent on mutual intra- and intercellular processes. The formation of such synchronising neuronal circuits or neuronal networks within the SCN is achieved by various mechanisms such as cell connections as well as humoral and paracrine factors.
Experiments on explanted and isolated SCN cells have led to the conclusion that in vivo cell compounds of the gap junction type mediate a significant proportion of synchronisation (Long et al. 2005). The neurotransmitters glutamate and γ-aminobutyric acid (GABA) are responsible for local intrinsic signalling and are distributed in almost the entire SCN (Albus et al. 2005). Glutamate is also used to transmit light information from the retinohypothalamic tract in the ventromedial SCN area, and GABAergic neurones project from the SCN to other regions of the hypothalamus, such as parts of the paraventricular nucleus.
In addition to GABA, several neuropeptides such as vasoactive intestinal peptide (VIP) expressed in the ventrolateral marginal zone and arginine vasopressin (AVP) produced in the dorsomedial nucleus are involved in information processing within the SCN and also in the regulation of hormone secretion synchronisation of oscillators located peripherally in the tissue (Hastings et al. 2007).
Through a combination of pharmacological and genetic manipulations, it has been shown in animal models that the circadian rhythm at the cellular level is partly determined by hierarchically arranged paracrine neuropeptidergic signalling. VIP plays a prominent role in this, supported by AVP and gastrin releasing peptide (GRP) (Maywood et al. 2006).
Afferent and efferent pathways of the SCN
The SCN has a large number of afferent and efferent connections. The above-mentioned RHT (retinohypothalamic tract) projects mainly into the ventrolateral area of the SCN, releases glutamate as well as pituitary adenylate cyclase activating peptide (PACAP) and substance P (SP) as neurotransmitters and thus transmits information about the ambient brightness for synchronisation with the light-dark alternation. Non-photonic information reaches the SCN by indirect innervations via the raphe nuclei and the intergeniculate leaflet (IGL) and thus contributes to the synchronisation of the SCN neurons (Hastings et al. 1996). The core area of the SCN also receives serotonin fibres from the raphe dorsal nucleus and the raphe magnus nucleus of the brainstem.
In addition to the afferent fibres mentioned above, a number of other brain regions project to the SCN, e.g. parts of the thalamus, the limbic system and the hypothalamus (Madeira et al. 2004).
Numerous brain regions are supplied with circadian control signals via a large number of direct and indirect efferent projections of the SCN (Fig. 1). Some connections, particularly in large parts of the hypothalamus and the projection to the pineal gland, which are involved in the control of complex neuroendocrine functions, have been characterised in more detail (Abrahamson and Moore 2001). Some examples are listed below:
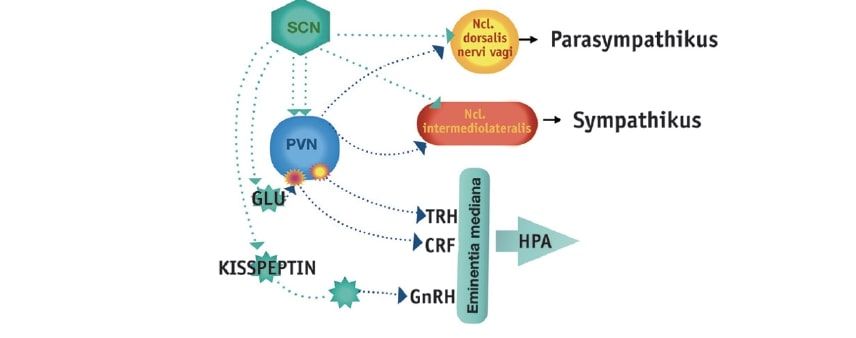
Fig. 1: Rhythm-generating neurones of the suprachiasmatic nucleus (SCN) project directly or via interneurones to the paraventricular nucleus (PVN) of the hypothalamus. After switching in the PVN, the projections reach the control instances of the hormonal axes (TRH = thyrotropin releasing hormone, CRF = corticotropin releasing factor, GnRH = gonadotropin releasing hormone) and the autonomic nervous system.
Hypothalamic-pituitary-adrenal axis ("hypothalamic-pituitary-adrenal axis"; HPA axis)
"Corticotropin releasing factor (CRF) (also known as corticotropin releasing hormone or corticoliberin) producing neurones of the paraventricular nucleus (PVN) of the hypothalamus are innervated by the SCN through direct projections or via interneurones, which causes the diurnal rhythmic release of CRF. In the diurnal state, the arginine vasopressin (AVP)-containing axons from the SCN promote the production of CRF and thereby activate the HPA axis (Ulrich-Lai et al. 2006).
The diurnal rhythmic control of cortisol secretion via the autonomous SCN-adrenal pathway is controlled by the sympathetic innervation of the adrenal glands (splanchnic nerve). It has been demonstrated in animal experiments that direct light information is associated with an increased release of cortisol. This occurs independently of the activation of the HPA axis (Fig. 2) (Chung et al. 2011)

Fig. 2: Daily rhythmic regulation of cortisol secretion and biosynthesis (based on Chung et al. 2011). Fluctuations in the circulating cortisol level are achieved by several regulatory mechanisms:
- Modulation of HPA axis activity by the SCN,
- Innervation of the splanchnic nerve of the adrenal cortex,
It has also been shown that in rats the diurnal rhythmic release of corticosterone (the most important steroid hormone in rodents) is influenced by modulation of the sensitivity of the adrenal glands to ACTH (Ulrich-Lai et al. 2006).
The sensitivity of the adrenal gland to ACTH stimulation is in turn regulated via the SCN-controlled sympathetic nerve innervation (splanchnic nerve) (Ulrich-Lai et al. 2006).
The molecular clockwork of the cortisol-producing cells of the adrenal cortex can also be influenced by the activation of the autonomous SCN-adrenal pathway (Oster et al. 2006; Ishida et al. 2005). Thus, the diurnal rhythmic release of cortisol is subject to direct SCN control by modulation of the HPA axis and by sympathetic splanchnic innervation of the adrenal gland. However, the local clockwork of the adrenal gland itself also plays an important role, e.g. in maintaining the cortisol rhythm by controlling the capacity and sensitivity of adrenal cortisol secretion and biosynthesis (Chung et al. 2011).
Clinical revelance
Dysregulated cortisol secretion can lead to numerous diseases and is associated with the development of Alzheimer's dementia and metabolic syndrome, among other things (Carroll et al. 2008; Ferrari et al. 2001; Pasquali et al. 2006; Bao et al. 2008). A decoupling of the fine-tuning between ACTH and cortisol secretion due to endogenous or exogenous influences has been associated in some studies with the occurrence of chronic fatigue syndrome, post-traumatic stress disorder, alcoholism and sepsis (De Kloet et al. 2006). In particular, various potentially disruptive influences such as shift/night work, jet lag, sleep deprivation or nocturnal food intake can result in severe dysregulation of circadian adaptation and the development of various pathologies, both short-term and chronic (Garaulet and Madrid 2009; Filipski et al. 2004).
Hypothalamic-pituitary-gonadal axis ("hypothalamic-pituitary-gonadal axis", HPG axis)
The SCN influences the gonadotropin releasing hormone (GnRH)-producing neurones in the nucleus praeopticus. In addition, circadian rhythms interact with peripheral serotoninergic systems to regulate lactation (Costa-e-Sousa and Hollenberg 2012). The neural oscillator in the SCN is essential for the timing of the pre-ovulatory rise in luteinising hormone (LH) (Kriegsfeld and Silver 2006; De la Iglesia and Schwartz 2006). In animal experiments, postsynaptic fibres of GnRH-producing cells in the SCN were identified as a correlate for the feedback regulation between the secretion of GnRH and the SCN (Van der Beek et al. 1997). Lesions of the SCN lead to impaired control of LH secretion, ovulation and the menstrual cycle in female rats (Wiegant and Terasawa 1982).
Clinical relevance
It is also clear from the regulation of the HPG axis that processes such as ovulation are not purely linear top-down processes, but are clocked by multioscillatory systems and depend on the normal functioning of the synchronisation of their coupled components (Sellix and Menaker 2010).
It is also clinically relevant that disturbances in the circadian rhythm of the HPG axis, e.g. during night/shift work or intercontinental flights, are involved in the development of infertility, menstrual disorders, menstrual pain, an altered duration of the follicular phase, altered FSH levels and follicular phases, low birth weights and an increased incidence of miscarriages (Fig. 3) (Mahoney 2010; Shechter and Boivin 2010).

Fig. 3: Hypothesis on a disturbed synchronisation of the HPG axis based on Sellix et al. (2010). Circadian oscillators are located in each component of the HPG axis. Disturbed synchronisation of the physiological processes is dependent on desynchronised relationships between the SCN, neurons of the GnRH, cells of the pituitary gland and ovarian cells. A disturbed synchronisation in the HPG axis can lead to a disturbance of the circadian clocks in target organs through a reduction in the amplitude or strength of the circadian clocks or a change in the amplitude or temporal structures of central or peripheral origin. desynchronisation in the HPG axis. This in turn can result in disorders of the reproductive organs and increase or even trigger reduced fertility (abbreviations: GnRH "gonadotropin releasing hormone"; LH luteinising hormone)
Thyrotropic control circuit (pituitary-thyroid control circuit, "hypothalamic-pituitary-thyroid axis", HPT axis)
The anterior part of the hypothalamus influences circadian variations of circulating thyrotropin (TSH) and thus the diurnal rhythmic fluctuations of peripheral thyroid hormones. This occurs by means of the projection of the SCN neurones into the parvocellular neurones of the paraventricular nucleus Kalsbeek et al. 2000; Costa-e-Sousa and Hollenberg 2012). However, thyroid hormones do not influence the circadian clock genes, so that the circadian rhythm reacts independently of negative feedback mechanisms, such as the inhibitory effect of free T4 on the secretion of the control hormone TSH Caria et al. 2009).
It is possible that the circadian regulation of the HPT axis is under the control of neuronal pathways, independent of negative feedback mechanisms of thyroid hormones. However, the possibility cannot be excluded that non-genomic processes of thyroid hormones control the excitability of neurons that form the hypothalamic circuitry and determine the rhythmicity of TRH release Costa-e-Sousa and Hollenberg 2012).
Clinical relevance
Clinically, the destruction of the SCN influences the rhythmic fluctuation of circulating corticosteroids, which in turn exert an inhibitory influence on TRH release (Alkemade et al. 2005). Accordingly, the HPA axis has an inhibitory effect on the HPT axis.
Neurovegetative (ANS)
Parasympathetic activity is regulated diurnally by means of SCN projections to the nucleus dorsalis nervi vagi. Sympathetic preganglionic neurones are influenced by connections to interneurones in the thoracic segment. In addition, the neurovegetative system is influenced via the SCN by the innervation of the preautonomous parvocellular neurones of the paraventricular nucleus. Axons that are localised dorsally and caudally in the paraventricular nucleus lead either directly - or via catecholaminergic neurons of the formatio reticularis - to the nucleus intermediolateralis of the thoracic section of the spinal cord and can thus influence the sympathetic preganglionic neurons, e.g. in the cervical ganglion superius.
Light stimulation directly increases the adrenaline level in the plasma (Ishida et al. 2005). As already mentioned, the circadian influence of the SCN on the HPA axis is mediated via the neurovegetative innervation of the adrenal glands (splanchnic nerve). Thus, light stimulation leads to an SCN-dependent rapid induction of Per1 expression in the adrenal gland (Ishida et al. 2005).
Epiphysis (pineal gland, corpus pineale, pineal organ)
The circadian control of the pineal gland via efferent pathways of the SCN is comparatively well studied. The endogenous oscillation generated in the SCN is modulated by RHT-mediated information about ambient light and projected to the paraventricular nucleus (PVN) of the hypothalamus. During the day there is GABAergic inhibition of the PVN neurones, whereas at night there is a glutamate-mediated excitation of the PVN neurones (Perreau-Lenz et al. 2003). The signal is transmitted from the PVN to the epiphysis via the intermediolateral nucleus (IML) of the spinal cord and the superior cervical ganglion (SCG). The epiphyseal hormone melatonin is synthesised and released at a high rate at night and at a low rate during the day.
In vitro studies on brain slices have identified melatonin receptors in the SCN (Vanecek et al. 1987), which enables a direct feedback of the released hormone to the circadian system and consecutively the shift of sleep-wake phases (Lewy et al. 1980). The nocturnal melatonin signal provides the time information for all cells and organs and, according to Blask (2009), is the most stable and reliable peripheral biomarker for measuring the time of the central biological clock (Fig. 4).

Fig. 4: Circadian control of the pineal gland: The neuronal pathway includes the paraventricular nucleus (PVN), nucleus intermediolateralis (IML), the ganglion cervicale superius (GCS) and the pineal gland
Sleep homeostasis is dependent on sleep pressure and the circadian rhythm. The ventrolateral preoptic nucleus (VLPO) of the anterior hypothalamus is a key factor for falling asleep and staying asleep, while the lateral-posterior part of the hypothalamus is partly responsible for waking behaviour (Saito et al. 2018).
A narrowly defined area of the lateral-posterior part of the hypothalamus contains specialised neurons that synthesise the neuropeptide orexin, which, in addition to its essential role in sleep-wake regulation, is also thought to play an important role in energy homeostasis and appetite formation.
The orexinergic pathways of the lateral-posterior part of the hypothalamus project into large parts of the brain, including the SCN, with most fibres ending in the area close to the SCN (Sakurai 2007). In the SCN itself, orexinergic fibres as well as a small amount of orexin secretion and orexin receptors have also been identified (Mondal et al. 1999). The latter were detected in particular on AVP (arginine vasopressin) and VIP (vasointestinal polypeptide) expressing cells (Bäckberg et al. 2002). The AVP- and VIP-expressing pathways in turn project to the orexinergic neurons in the lateral-posterior part of the hypothalamus (Abrahamson and Moore 2001), which, in addition to a close morphological relationship, also suggests a functional link between the lateral-posterior part of the hypothalamus and the circadian network of the SCN. This is expressed in the diurnally fluctuating orexin level in the cerebrospinal fluid (Grady et al. 2006).
Of practical relevance is also the suspicion that wearing sunglasses in the morning, especially with orange-coloured lenses that block blue light, hinders the resynchronisation of the biological clock by sunlight (Sasseville et al. 2006).
Clocks everywhere: the organism's circadian clock network
As explained in the section on circadian regulation of the neurohormonal axes, although the SCN can be regarded as the central pacemaker, local oscillatory systems at organ and tissue level also play an important role in circadian regulation. These locally effective circadian mechanisms have been investigated many times in experiments. However, circadian rhythms persist even without external light information and social influences (Czeisler and Klerman 1999).
Animal experiments have shown that circadian clocks occur in practically every cell and every organ tissue and are synchronised by the SCN under normal conditions (Buijs and Kalsbeek 2001; Balsalobre 2002).
In the event of a time shift and the resulting desynchronisation between the internal and external time structure, the central pacemaker adjusts comparatively quickly (Yamazaki al. 2000). The peripheral clocks of the internal organs, however, adapt to the new temporal framework at different speeds and to different degrees, which leads to a decoupling of the individual rhythms in the organism's circadian network with the resulting symptoms (Hastings et al. 2003).
Another aspect of the complexity of the endogenous time programme lies in the different response behaviour of central and peripheral clocks with regard to external timing stimuli. For example, SCN reacts to light, whereas the internal clock of the liver reacts primarily to food intake: like other peripheral oscillators, it is SCN-controlled and only indirectly subject to the influence of light (Damiola et al. 2000; Stokkan et al. 2001).
Peripheral internal clocks have existed for over 600 million years. Even fruit flies have them. Every cell and every organ has its own clocks, e.g. 3-20% of the genes have a rhythmic expression. 12 different clock genes and around 20 modulating genes have been identified in humans to date.
In the search for the genetic basis of biological clocks, a clock gene was first found in the fruit fly Drosophila. If the molecular mechanism of the intrinsic oscillator was disturbed by a targeted intervention in the genetic material of the fly, it presented arrhythmic activity phases, or these were completely absent (Konopka and Benzer 1971). In the 1990s, the first clock gene was identified in mammals (mice). This was followed by numerous other genes and gene products whose diurnal rhythmic expression reacts to timing stimuli (Honma et al. 2004; Shearman et al. 2000).
According to the current genetic model of the mammalian circadian clock, interlocking transcriptional-translational feedback (TTL) loops form the basis for circadian oscillation at the cellular level (Brown et al. 2012).
Briefly summarised, during the day the transcription factor CLOCK ("circadian locomotor output cycles caput") or NPAS2 ("neuronal PAS domain-containing protein 2") is bound with BMAL1 ("brain and muscle aryl hydrocarbon receptor nuclear translocator-like 1") to E-box ("enhancer box") promoters to stimulate the unfolding of period (Per1-3) and cryptochrome (Cry1/2) and others. This results in an increase of PER/CRY protein complexes in the cytoplasm during the day, which later move to the nucleus where they inhibit the activity of CLOCK-BMAL1 complexes (or NPAS2-BMAL1 complexes). This leads to a switch-off of Per/Cry transcription during the night. Towards the next morning, as the PER/CRY complexes in the cell nucleus are increasingly degraded, a new cycle begins with an increase in PER/CRY protein complexes in the cytoplasm (Dumbell and Matveeva 2016).
In addition to photoreceptors, circadian genes (PER2, 3 or CLOCK) are explicitly associated with the regulation of cell ageing and uncontrolled cell growth (Fu and Lee 2003). For example, a defect in the period gene leads to a loss of peripheral rhythm. A molecular biological deletion of a single gene controlling the circadian rhythm (PER2) in animal experiments causes the affected animals to age and die dramatically faster than the comparison group of genetically identical rats with intact rhythm genes (Fu and Lee 2003; Lee 2006). Peripheral circadian rhythms exist, for example, in the gene expression of liver, adipose tissue, muscles, mammary gland and heart tissue (Green et al. 2008).
These internal clocks are synchronised and show a cycle of 24.20 hours in humans, but are relatively inaccurate compared to the SCN. The continuous synchronisation of internal cellular time measurements and the resulting possible self-correction of gene and protein expression with external time cues, in particular light information by means of the SCN, but also through social factors, results in precise rhythmic coordination in the organism (Pittendrigh 1993).
On the one hand, the partial flexibility of the self-excited biological oscillator allows a harmonious temporal coordination of the physiology and behaviour of an organism with its externally or internally induced changes. On the other hand, it must remain stably synchronised with the external day in order to be able to anticipate periodic processes in the environment. Every observed circadian rhythm is the product of endogenous and exogenous influences (Fig. 5).

Fig. 5: Synchronisation of circadian time control in the body. (Modified from Hastings et al. 2007). Circadian coordination within the organism in which the primary pacemaker of the SCN is maintained and synchronised by solar radiation from retinal afferents, tissue-based clocks in the major organ systems and by interactions of endocrine, neurovegetative, behavioural (nutritional) and immunological signals
Clinical relevance of the findings on the circadian rhythm
Chronically ill people often appear to have shifts in their circadian rhythm (Kanikowska et al. 2015; Moore-Ede et al. 1983; Luce 1970). For example, changes in sleep cycles impair the immune system (Bollinger et al. 2010).
The clinical manifestation of shifts in the circadian rhythm could therefore be taken into account in osteopathic treatment, especially for chronic diseases. However, circadian rhythm considerations also play a major role in the therapeutic support of healthy individuals, for example athletes in relation to sleep-wake behaviour, jet lag during international competitions, the times of food intake, training routines and types of exercise, etc. Drust et al. 2005; Hammouda et al. 2013).
Differences in time-dependent peak performance can be attributed to internal physiological mechanisms and circadian influences Facer-Childs and Brandstaetter 2015). For example, jet lag caused by travelling more than 5 hours in different time zones increases susceptibility to illness by a factor of 2-3 Schwellnus et al. 2012).
Many people, especially the late chronotype (so-called owl phenotype), experience daily repetitive circadian disruptions that can affect performance, health and well-being Horne and Moseley 2011; Roenneberg et al. 2013; Samuels 2009).
Conclusion
Living organisms present an endogenous periodicity in behaviour (sleeping and waking, rest and activity, attention) and physiology (body temperature, hormone release, metabolic processes, etc.). They therefore not only adapt to changes in the environment reflexively, but also anticipate them in order to be optimally prepared. The ability to anticipate environmental conditions and processes is an essential mechanism for the successful survival of a living organism and has led to the development of endogenous time programmes in the course of evolution.
The morphological correlate of the endogenous time programme in mammals is the suprachiasmatic nucleus (SCN) in the anterior hypothalamus. The intrinsic molecular clockwork generates a self-sustaining oscillation with a period of approximately one day (circadian) and is synchronised by stable external stimuli from the environment (so-called zeitgebers). The most important zeitgeber for living organisms is (sun)light or the day-night cycle. The SCN receives information about the intensity and spectral composition of the incident light via specialised retinal ganglion cells and the retinohypothalamic tract (RHT). The neurons of the SCN are synchronised with each other by means of paracrine mechanisms and transmit circadian information to central and peripheral effectors. The circadian network in the SCN thus integrates and regulates both the external environmental stimuli and the internal temporal signals of the peripheral organ and tissue oscillators.
Chronically ill people in particular often show shifts in their biological rhythms. Taking these chronobiological shifts into account is an important aspect in the treatment of chronic illnesses. In the osteopathic context, this is less about palpable rhythmic phenomena, as speculatively formulated in cranial or visceral aspects of osteopathy. Rather, the focus here is on an understanding of polyrhythmic phenomena and interactions. In this sense, symptoms are proactive signs that can be understood as an indication of a change in behaviour. Accordingly, it is less about trying to manipulate brain centres palpatorily, for example, and more about supporting the patient in perceiving, learning to read and understand chronobiological dysfunctions and desynchronisation between the internal and external time structure and initiating targeted behavioural changes or avoidance behaviour in order to improve homeostasis. For example, how can I recognise at an early stage, ideally before chronic complaints arise, that my stress axis has been activated and is affecting my rhythmic patterns, such as sleep, and how can I react and act appropriately?
The aim here is to support the patient's personal responsibility. Psychoeducation and learning experiences that are important for one's own life are therefore also essential aspects of osteopathic interactions. Osteopathic manipulative palpations can support this self-regulation and train the patient's own body awareness.
Literature
[1] Abrahamson EE, Moore RY. Suprachiasmatic nucleus in the mouse: retinal innervation, intrinsic organisation and efferent projections. Brain Res 2001 916: 172-191
[2] Albus H, Vansteensel MJ, Michel S, Block GD, Meijer JH, 2005. A GABAergic mechanism is necessary for coupling dissociable ventral and dorsal regional oscillators within the circadian clock. Curr Biol 2005; 15: 886-893
[3] Alkemade A, Unmehopa UA, Wiersinga WM, Swaab DF, Fliers E. Glucocorticoids decrease thyrotropin-releasing hormone messenger ribonucleic acid expression in the paraventricular nucleus of the human hypothalamus. J Clin Endocrinol Metab 2005; 90: 323-327
[4] Bäckberg M, Hervieu G, Wilson S, Meister B. Orexin receptor-1 (OX-R1) immunoreactivity in chemically identified neurons of the hypothalamus: focus on orexin targets involved in control of food and water intake. Eur J Neurosc 2002; 15: 315-328
[5] Balsalobre A. Clock genes in mammalian peripheral tissues. Cell Tissue Res 200; 309 (1): 193-9
[6] Bao AM, Meynen G, Swaab DF, The stress system in depression and neurodegeneration: focus on the human hypothalamus, Brain Res. Rev 2008; 57: 531-553
[7] Blask DE. Melatonin, sleep disturbance and cancer risk. Sleep Medicine Reviews. 2009; 13: 257-264
[8] Bollinger T, Bollinger A, Oster H et al. Sleep, immunity, and circadian clocks: a mechanistic model. Gerontology 2010; 56 (6): 574-80
[9] Brown SA, Kowalska E, Dallmann R. (Re)inventing the circadian feedback loop. Dev Cell 2012; 22 (3): 477-87
[10] Buijs RM and A. Kalsbeek. Hypothalamic integration of central and peripheral clocks. Nat Rev Neurosci 2001; 2 (7): 521-6
[11] Caria MA, Dratman MB, Kow LM, Mameli O, Pavlides C. Thyroid hormone action: Nongenomic modulation of neuronal excitability in the hippocampus. J Neuroendocrinol 2009; 21: 98-107
[12] Carroll T, Raff H, Findling JW. Late-night salivary cortisol measurement in the diagnosis of Cushing's syndrome. Nat Clin Pract Endocrinol Metab 2008; 4: 344-350
[13] Chung S, Son GH, Kim K. Circadian rhythm of adrenal glucocorticoid: its regulation and clinical implications. Biochim Biophys Acta 2011; 1812 (5): 581-591
[14] Costa-e-Sousa RH, Hollenberg AN. Minireview: The Neural Regulation of the Hypothalamic-Pituitary-Thyroid Axis. Endocrinology 2012; 153 (9): 4128-4135
[15] Czeisler CA, Shanahan TL et al. Suppression of melatonin secretion in some blind patients by exposure to bright light. N Engl J Med 1995; 332 (1): 6-11
[16] Czeisler CA, Klerman EB. Circadian and sleep-dependent regulation of hormone release in humans. Recent Prog Hormone Res 1999; 54: 97-130 (discussion 130-132)
[17] Dacey DM et al. Melanopsin-expressing ganglion cells in primate retina signal colour and irradiance and project tot he LGN. Nature 2005; 433, 749-754
[18] Damiola, F, N. Le Minh et al. Restricted feeding uncouples circadian oscillators in peripheral tissues from the central pacemaker in the suprachiasmatic nucleus. Genes Dev 2000; 14 (23): 2950-61
[19] De Kloet CS, Vermetten E, Geuze E et al. Assessment of HPA-axis function in post-traumatic stress disorder: pharmacological and non-pharmacological challenge tests, a review J Psychiatr Res 2006; 40: 550-567
[20] De la Iglesia HO, Schwartz WJ. Minireview: timely ovulation: circadian regulation of the female hypothalamo-pituitarygonadal axis. Endocrinology 2006; 147: 1148-1153
[21] Dibner C, Schibler U, Albrecht U. The mammalian circadian timing system: organisation and coordination of central and peripheral clocks. Annu Rev Physiol 2010; 72: 517-49
[22] Drust B, Waterhouse J, Atkinson G. Circadian rhythms in sports performance-an update. Chronobiology International 2005; 22 (1): 21-44
[23] Dumbell R, Matveeva O, Oster H. Circadian clocks, stress, and immunity. Front Endocrinol 2016; 7: 37
[24] Facer-Childs E, Brandstaetter R, The impact of circadian phenotype and time since awakening on diurnal performance in athletes. Curr Biol 2015; 25: 1-5
[25] Ferrari E, Casarotti B, Muzzoni B et al. Age-related changes of the adrenal secretory pattern: possible role in pathological brain aging. Brain Res Rev 2001; 37: 294-300
[26] Filipski E, Delaunay F, King VM et al. Effects of chronic jet lag on tumour progression in mice. Cancer Res 2004; 64: 7879-7885
[27] Foster RG, Hankins MW et al. Non-rod, non-cone photoreception in the vertebrates. Prog Retin Eye Res 2002; 21 (6): 507-27
[28] Foster RG. Neurobiology: bright blue times. Nature 2005; 433 (7027): 698-9
[29] Fu L, Lee CC. The circadian clock: pacemaker and tumour suppressor. Nat Rev Cancer 2003; 3 (5): 350-361
[30] Garaulet M, Madrid JA. Chronobiology, genetics and metabolic syndrome. Curr Opin Lipidol 2009; 20 (2): 127-34
[31] Gaston, S, Menaker, M et al. Pineal function: the biological clock in the sparrow? Science 1968; 160 (832): 1125-7
[32] Gharbi A, Masmoudi L, Ghorbel S. Time of day effect on soccer-specific field tests in Tunisian boy players. Adv Phys Educ 2013; 3, 2: 71-75
[33] Grady SP, Nishino S, Czeisler CA, Hepner D, Scammell TE. Diurnal variation in CSF Orexin-A in healthy male subjects. Sleep 2006; 29: 295-297
[34] Green CB, Takahashi JS, Bass J. The Meter of Metabolism. Cell 2008; 134 (5): 728-742
[35] Hammouda O et al. Time-of-day effects on biochemical responses to soccer-specific endurance in elite Tunisian football players. J Sports Sci 2013; (31) 9: 963-971
[36] Hastings MH, Best JD, Ebling FJP, Maywood ES, McNulty S, Schurov I, Selvage D, Sloper P, Smith KL. Entrainment of the circadian clock. Hypothalamic Integrat Circad Rhyth 1996; 111: 147-174
[37] Hastings MH, Reddy AB et al. Expression of clock gene products in the suprachiasmatic nucleus in relation to circadian behaviour. Novartis Found Symp 2003; 253: 203-17; discussion 102-9, 218-22, 281-4
[38] Hastings M, O'Neill JS, Maywood ES. Circadian clocks: regulators of endocrine and metabolic rhythms. J Endocrinol 2007; 195: 187-198
[39] Herzog ED, Aton SJ, Numano R, Sakaki Y, Tei H. Temporal precision in the mammalian circadian system: A reliable clock from less reliable neurons. J Biol Rhyth 2004; 19: 35-46
[40] Honma S, Nakamura W, Shirakawa T, Honma K Diversity in the circadian periods of single neurons of the rat suprachiasmatic nucleus depends on nuclear structure and intrinsic period. Neurosc Lett 2004; 358: 173-176
[41] Horne J, Moseley R. Sudden early-morning awakening impairs immediate tactical planning in a changing 'emergency' scenario. J Sleep Res 2011; 20: 275-278
[42] Inouye ST, Kawamura H et al. Persistence of circadian rhythmicity in a mammalian hypothalamic 'island' containing the suprachiasmatic nucleus. Proc Natl Acad Sci USA 1979; 76 (11): 5962-6
[43] Ishida A, Mutoh T, Ueyama T et al. Light activates the adrenal gland: timing of gene expression and glucocorticoid release. Cell Metab 2005; 2: 297-307
[44] Kalsbeek A, Fliers E, Franke AN, Wortel J, Buijs RM. Functional connections between the supreachiasmatic nucleus and the thyroid gland as revealed by lesioning and viral tracing techniques in the rat. Endocrinology 2000; 141: 3832-3841
[45] Kanikowska D, Sato M, Witowski J. Contribution of daily and seasonal biorhythms to obesity in humans. Int J Biometeorol 2015; 59 (4): 377-384
[46] Konopka RJ, Benzer S. Clock mutants of Drosophila melanogaster. Proc Natl Acad Sci USA 1971; 68: 2112-2116
[47] Kriegsfeld LJ, Silver R. The regulation of neuroendocrine function: Timing is everything. Horm Behav 2006; 49: 557-574
[48] Lee CC. Tumour suppression by the mammalian Period genes. Cancer Causes Control 2006; 17 (4): 525-530
[49] Lewy A J, T. A. Wehr et al. Light suppresses melatonin secretion in humans. Science 1980; 210 (4475): 1267-9
[50] Long MA, Jutras MJ, Connors BW, Burwell RD. Electrical synapses coordinate activity in the suprachiasmatic nucleus. Nat Neurosci 2005; 8: 61-66
[51] Lucas R J, Foster R G et al. Photoentrainment in mammals: a role for cryptochrome? J Biol Rhyth 1999; 14 (1): 4-10
[52] Luce GG. Biological rhythms in psychology and medicine. Maryland, NIMH; 1970
[53] Madeira MD, Pereira PA, Silva SM, Cadete-Leite A, Paula-Barbosa MM, 2004. Basal forebrain neurons modulate the synthesis and expression of neuropeptides in the rat suprachiasmatic nucleus. Neuroscience 2004; 125: 889-901
[54] Mahoney MM. Shift work, jet lag, and female reproduction. international J Endocrinol 2010; 813764
[55] Maywood ES, Reddy AB, Wong GKY, O'Neill JS, O'Brien JA, McMahon DG, Harmar AJ, Okamura H, Hastings MH. Synchronisation and maintenance of timekeeping in suprachiasmatic circadian clock cells by neuropeptidergic signalling. Curr Biol 2006; 16: 599- 605
[56] Mondal MS, Nakazato M, Date Y, Murakami N, Hanada R, Sakata T, Matsukura S. Characterisation of orexin-A and orexin-B in the microdissected rat brain nuclei and their contents in two obese rat models. Neuroscience Letters 1999; 273: 45-48
[57] Moore RY, Eichler V. B et al. Loss of a circadian adrenal corticosterone rhythm following suprachiasmatic lesions in the rat. Brain Res 1972; 42 (1): 201-6
[58] Moore-Ede MC, Czeisler CA, Richardson GS. Circadian timekeeping in health and disease. New Engl J Med 1983; 309: 469-536
[59] Oster H, Damerow S, Kiessling S et al. The circadian rhythm of glucocorticoids is regulated by a gating mechanism residing in the adrenal cortical clock. Cell Metab 2006; 4: 163-173
[60] Panda S, Sato T K et al. Melanopsin (Opn4) requirement for normal light-induced circadian phase shifting. Science 2002; 298 (5601): 2213-2216
[61] Pasquali R, Vicennati V, Cacciari M, Pagotto U. The hypothalamic - pituitary - adrenal axis activity in obesity and the metabolic syndrome. Ann NY Acad Sci 2006; 1083: 111-128
[62] Perreau-Lenz S, Kalsbeek A, Garidou ML, Wortel J, van der Vliet J, van Heijningen C, Simonneaux V, Pevet P, Buijs RM. Suprachiasmatic control of melatonin synthesis in rats: inhibitory and stimulatory mechanisms. European Journal of Neuroscience 2003; 17: 221-228
[63] Pevet P. Melatonin and biological rhythms. Biol Signals Recept 2000; 9 (3-4): 203-212
[64] Pittendrigh CS. Temporal organisation: reflections of a Darwinian clockwatcher. Annu Rev Physiol 1993; 55: 16-54
[65] Provencio, I, Rollag MD et al. Photoreceptive net in the mammalian retina. This mesh of cells may explain how some blind mice can still tell day from night. Nature 2002; 415 (6871): 493
[66] Ralph MR, Menaker M. A mutation of the circadian system in golden hamsters. Science 1988; 241 (4870): 1225-7.s
[67] Richter CP. Sleep and activity: their relation to the 24-hour clock. Res Publ Assoc Res Nerv Ment Dis 1967; 45: 8-29
[68] Roenneberg T, Kantermann T, Juda M et al. Light and the human circadian clock. Handbook Exp Pharmacol 2013; 217: 311-331
[69] Saito YC, Maejima T, Nishitani M, Hasegawa E, Yanagawa Y, Mieda M, Sakurai T. Monoamines Inhibit GABAergic Neurons in Ventrolateral Preoptic Area That Make Direct Synaptic Connections to Hypothalamic Arousal Neurons. J Neurosci 2018; 11: 2835-2817
[70] Sakurai T. The neural circuit of orexin (hypocretin): maintaining sleep and wakefulness. Nature Reviews Neuroscience 200/, 8: 171-181
[71] Samuels C. Sleep, recovery, and performance: the new frontier in high-performance athletics. Phys Med Rehabil Clin North Am 2009; 20: 149-159, ix
[72] Sasseville A, Paquet N, Sevigny J, Herbert M. Blue blocker glasses impede the capacity of bright light to suppress melatonin production. J. Pineal Res 2006; 41: 73-78
[73] Schwartz WJ, Davidsen LC et al. In vivo metabolic activity of a putative circadian oscillator, the rat suprachiasmatic nucleus. J Comp Neurol 1980; 189 (1): 157-67
[74] Schwellnus MP, Derman WE, Jordaan E et al. Elite athletes travelling to international destinations >5 time zone differences from their home country have a 2-3-fold increased risk of illness Br J Sports Med 2012; 46: 816-821
[75] Sekaran, S, Foster RG et al. Calcium imaging reveals a network of intrinsically light-sensitive inner-retinal neurons. Curr Biol 2003; 13 (15): 1290-8
[76] Sellix MT, Menaker M. Circadian Clocks in the Ovary. Trends Endocrinol Metab (TEM) 2010; 21 (10): 628-636
[77] Shearman LP, Sriram S, Weaver DR, Maywood ES, Chaves I, Zheng BH, Kume K, Lee CC, van der Horst GTJ, Hastings MH, Reppert SM. Interacting molecular loops in the mammalian circadian clock. Science 2000; 288: 1013-1019
[78] Shechter A, Boivin DB. Sleep, hormones, and circadian rhythms throughout the menstrual cycle in healthy women and women with premenstrual dysphoric disorder. Int J Endocrinol 2010; 259345
[79] Stephan FK, Zucker I,. Circadian rhythms in drinking behaviour and locomotor activity of rats are eliminated by hypothalamic lesions. Proc Natl Acad Sci USA 1972; 69 (6): 1583-6
[80] Stokkan K A, S. Yamazaki et al. Entrainment of the circadian clock in the liver by feeding. Science 291 (5503): 490-3, Storch K F, O. Lipan et al. Extensive and divergent circadian gene expression in liver and heart. Nature 2001; 417 (6884): 78-83
[81] Storch KF, Lipan O et al. Extensive and divergent circadian gene expression in liver and heart. Nature 2002; 417 (6884): 78-83
[82] Ulrich-Lai YM, Arnhold MM, Engeland WC. Adrenal splanchnic innervation contributes to the diurnal rhythm of plasma corticosterone in rats by modulating adrenal sensitivity to ACTH, Am J Physiol Regul Integr Comp Physiol 2006; 290: R1128-R1135
[83] Underwood, H, R. K. Barrett et al. Melatonin does not link the eyes to the rest of the circadian system in quail: a neural pathway is involved. J Biol Rhythms 1990; 5 (4): 349-61
[84] Van der Beek EM, Wiegant VM, van Oudheusden HJC, van der Don HA, van den Hurk R, Buijs RM. Synaptic contacts between gonadotropin-releasing hormone-containing fibres and neurons in the suprachiasmatic nucleus and perichiasmatic area: an anatomical substrate for feedback regulation?. Brain Res 1997; 755 (1): 101-111
[85] Vanecek J, Pavlik A, Illnerova H. Hypothalamic melatonin receptor sites revealed by autoradiography. Brain Res 1987; 435: 359-362,
[86] Welsh D K, Logothetis DE et al. Individual neurons dissociated from rat suprachiasmatic nucleus express independently phased circadian firing rhythms. Neuron 1995; 14 (4): 697-706
[87] Wiegant SJ, Terasawa E. Discrete lesions reveal functional heterogeneity of suprachiasmatic structures in regulation of gonadotropin secretion in the female rat. Neuroendocrinology 1982; 34: 395-404
[88] Yamazaki, S, R. Numano et al. Resetting central and peripheral circadian oscillators in transgenic rats. Science 2000; 288 (5466): 682-5



